Quality Assurance
Policy
Products and services provided by the Asahi Kasei Group internally and externally include materials, products, installations, various services, and after-sales support. We believe that providing safe and reliable products and services that satisfy our customers is our ultimate mission.
We have established the Asahi Kasei Group Quality Policy and Regulation on Quality Assurance, and based on these, we promote quality assurance to provide products and services that satisfy our customers and society.
Asahi Kasei Group Quality Policy
The Asahi Kasei Group flexibly anticipates the constantly changing needs of customers and society to create and provide products and services with quality that ensures safety and security.
Management Framework
In April 2019, we appointed a dedicated Executive Officer for Quality Assurance to further reinforce the management framework.
The Corporate Quality Assurance department of Asahi Kasei Corporation oversees and coordinates Group-wide quality assurance activities.
Corporate Quality Assurance consists of three groups: the Corporate Quality Assurance Planning Group, which proposes new plans and supports quality assurance activities at each internal organization; the Corporate Quality Audit Group, which proposes improvements for preventing compliance risks that occur during quality assurance activities at business organizations and offers support in preparing governance; and the Corporate Chemical Regulatory Group, which fulfills the function of ensuring compliance with chemical laws and regulations as a diversified chemical manufacturer. Corporate Quality Assurance performs a head-office function as a hub for the Group's quality assurance framework and strives every day to reinforce quality assurance by coordinating activities across the entire Asahi Kasei Group to deliver safe and reliable products and services to our customers and society.
Each core operating company and strategic business unit within the Asahi Kasei Group performs quality assurance in accordance with the products and services provided in each business area, in conformity with the Group Principles.
Regulation on Quality Assurance stipulate the quality assurance activities to be led by Asahi Kasei ESH & QA Manager, such as the presidents of the core operating companies and strategic business units. The bylaws also define the appointment and roles of Quality Assurance Managers who play a central role in activities to enhance quality assurance in the relevant organization. The Quality Assurance Managers' Conference is held four times a year to transmit and share information among the entire Asahi Kasei Group regarding quality assurance activities. In addition, from fiscal 2019, we have started to provide an opportunity for the Executive Officer for Quality Assurance and the Senior General Manager of Corporate Quality Assurance to meet directly with ESH & QA Managers, Quality Assurance Managers, and others to discuss the enhancement of quality assurance and for frank exchange of opinions and sharing of ideas through face-to-face meetings, thereby creating an environment that enables us to build a quality assurance system with reliable safety and security.
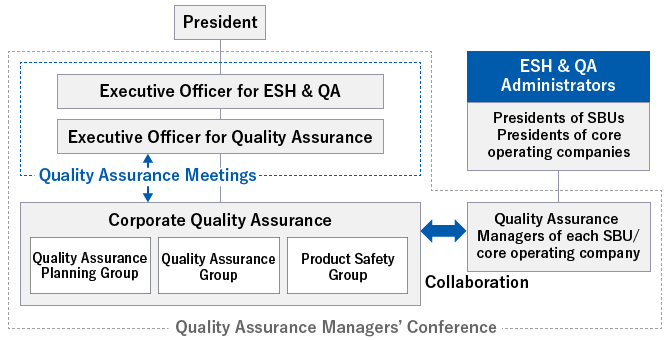 Diagram of quality assurance framework
Diagram of quality assurance framework- Quality Assurance Meeting (held each month):
- Based on the Monthly Quality Assurance Report prepared by Corporate Quality Assurance, the Executive Officer for ESH & QA, the Executive Officer for Quality Assurance, and the Senior General Manager of Corporate Quality Assurance of Asahi Kasei Corporation hold the Quality Assurance Meeting to discuss information related to quality assurance.
- Quality Assurance Managers' Conference (held four times a year):
- Quality Assurance Managers, who play a central role in strengthening quality assurance, meet to transmit and share information among the entire Asahi Kasei Group.
Quality assurance initiatives
The Asahi Kasei Group defines the procedure for risk assessment in quality assurance by internal company regulations.
Risk assessment is implemented at each stage of product development and product supply, to ensure product safety and to prevent product problems and complaints from emerging. In addition, we have also established appropriate measures to be taken if a serious product problem or serious accident occurs or is likely to occur.
![[Product development phase] [Proposal] Confirmation of implementation content and schedule to ensure safety of products and services
(Ex.)
・Designation of applications and destinations
・Researching regulation and standards
・Researching hazards
→ [Pre market-evaluation study] Evaluation of safety and risks, Elimination and reduction of risks, Identification of risks for users and risk reduction measures, Preparation of documentation, Identification of conditions for production and distribution → [Pre market-evaluation safety review] → [Pre-marketing study] Evaluation of safety and risks, Elimination and reduction of risks, Understanding methods of use and disposal, Identification of risks for users and risk reduction measures, Preparation of documentation, Confirmation and revision of conditions for production and distribution → [Pre-marketing safety review] → [Product supply phase] [Post-marketing study] Understanding methods of use and disposal, Revision of documentation, Application of conditions for production and distribution](/sustainability/social/quality/images/index-img-03.png) Flow of quality assurance activities
Flow of quality assurance activities
Chemical substance management
The Asahi Kasei Group identifies the properties of chemical substances and appropriately manages each process from product development, raw material procurement, and production (including intermediates) through to use and disposal in order to ensure the safety of products and production processes. We implement the chemical substance management shown in the diagram below at each stage from the perspectives of the global environment, process safety, occupational health and safety/physical health, and quality assurance.
The Asahi Kasei Group implements chemical substance management focused on each business division in cooperation with Corporate Quality Assurance and Corporate ESH of Asahi Kasei Corporation.
-
![Chemical substance management flow Society / Asahi Kasei Group / Green procurement, Global environment: Reduction of environmental burden, Process safety: Prevention of fires and explosions, Physical distribution safety: Provision of Transport Emergency Cards / Supplier: [Supply]→[Material]→[Procurement]→[Production][R&D]→[Sale]→ Customer: [Product]→[Use]→[Disposal] / Receipt of SDSs for materials, Occupational health and safety: Prevention of workplace injuries, Product safety: Distribution of SDSs for products,](/sustainability/social/quality/images/index-img-06.png) Chemical substance management flow
Chemical substance management flow
R&D
The management of chemical substances begins in the R&D stage when the applications for chemical substances are determined, and from that stage onwards we strive to design products and process while being considerate of the environment. For products that are expected to be exported to other countries in the future in addition to being sold domestically, we conduct research on each country’s laws and regulations and consider the requisite measures.
Materials purchase
When purchasing materials, information related to the safety of chemical substances is received from the supplier. This information serves as a guide to safe storage and handling.
Production
At the production stage, we manage chemical substances, including intermediates, in an appropriate manner to suppress emissions into the environment. We also strive to prevent fires, explosions, and leaks at facilities where chemical substances are handled to ensure the safety of local communities and preserve the global environment. To protect the health of employees, we manage chemical substances by performing sound risk assessment for chemical substances and preventing workplace exposure to hazardous substances.
Sale, use, and disposal
Guidance for proper sale, use and disposal of chemical substances and chemical products is provided in Safety Data Sheets (SDSs), technical bulletins, and product brochures.
Transport Emergency Cards are issued to guide the proper environmental and safety response in the event of an accident during physical distribution. Moreover, when products are exported outside of Japan, we take appropriate measures to comply with local laws and regulations.
Appropriate labeling and information provision
As the Asahi Kasei Group provides customers with products and services that are end products for domestic and household-use products, we provide information that includes product performance, precautions, and suggested usage to ensure safe use of our products and services.
We endeavor to provide descriptions of products and services to customers, including product labeling and advertisements, that are easy to understand and not misleading. In addition, we confirm the content of descriptions and advertisements of products and services at each stage from product development and introduction to sale, and continuously check that there is no infringement of related laws, regulations, or voluntary industry standards, and confirm that customers are able to properly use products and services safely and reliably.
Compliance with chemical substance regulations around the world and sharing of information
As laws and regulations concerning chemical substances continue to be adopted around the world, we have been confirming, responding to, and managing them within the company to ensure compliance. In addition, some of these regulations require the sharing of information. Besides providing the necessary information to our customers, we actively work to provide information on chemical substances contained in products throughout the supply chain. One of our activities is participating in the Joint Article Management Promotion-consortium (JAMP) as an upstream company since its establishment in 2008. JAMP is promoting the dissemination of information on such chemical substances. We continue to promote the use of chemSHERPA, a communication tool, as part of JAMP’s activities.
Responding to Globally Harmonized System (GHS)
Globally Harmonized System of Classification and Labelling of Chemicals (GHS) is a system for classifying and labelling chemicals by hazard in accordance with globally unified rules in order to help with accident prevention and health and environmental protection. The Asahi Kasei Group is advancing a program to classify the hazards of all of our chemical products in accordance with GHS categories, and revise our SDSs and label our products with safety information accordingly.
Quality assurance training and human resources development
Development of core human resources for quality assurance
We have held the Quality Assurance Forum since fiscal 2017 to foster core human resources for quality assurance in the Asahi Kasei Group. In fiscal 2024, 45 employees selected from throughout the Asahi Kasei Group gathered once per month for a period of six months for group discussions on a certain subject each time, combined with lectures on each subject by outside experts (mainly university professors at the forefront of the Japanese Society for Quality Control and experts who gained practical experience in quality assurance in companies). At the final session, participants in each business sector discuss issues in their own organizations based on what they learned, and prepare a declaration summarizing their own future actions for organizational improvement.
In fiscal 2025, we are continuing this as a key project for in-house quality assurance training.
Fostering a quality assurance mindset
In fiscal 2024, we worked to foster a quality assurance mindset primarily through the following three activities.
- Quality education
These activities featured the Executive Officer for Quality Assurance independently visiting various sites in Japan and overseas and giving lectures to gain each individual’s understanding and eliminate quality incidents by raising the capabilities of our workplaces. Under the policy that “Everyone has a role in quality,” we implemented these activities for not only the plants, but also sales, administrative personnel, and management separately.
- Town hall meetings
With the aims of building a relationship of mutual understanding and trust between the Executive Officer for Quality Assurance and the workers on the front lines of the workplace, and creating a foundation that enables each individual worker to be active and fulfilled, we hold Asahi Kasei town hall meetings with lectures and workshops at various sites in Japan and overseas. Over 3,000 employees have participated in these meetings at 62 sites in Japan and overseas (as of May 2025).
- Monthly quality activities
Coinciding with Quality Month in November, the President of Asahi Kasei, the Presidents of core operating companies, the president of each strategic business unit, and the Senior General Manager of Corporate Quality Assurance issued messages to Group employees about the importance of quality assurance. Training via e-learning was conducted as well to raise awareness on the subject.
These activities will be continued in fiscal 2025 and beyond, as we look to devote even more effort to fostering human resources through expansion of robust quality education.
Education on compliance with chemical substance regulations
We provide regular training to research, production, and sales staff in each region where we are located. Such training includes sharing the most up-to-date information on the latest chemical substance-related laws and regulations (Act on the Evaluation of Chemical Substances and Regulation of Their Manufacture, etc., Industrial Safety and Health Act, Poisonous and Deleterious Substances Control Act, etc.) in Japan and overseas for the compliance with chemical substance regulations, examining possible responses, and presenting the latest themes in chemical substance management.
Mechanisms to utilize customer feedback
We believe satisfying customers and providing products and services that are a delight to use translates into contribution to society. In order to achieve this, we believe that it is most important to identify true needs by listening carefully to customer feedback to establish two-way communication. We have built frameworks for such communication with customers in each of our businesses and strives to listen to frank and honest feedback.
![[Materials, intermediates, devices] [Asahi Kasei Group] Sales and R&D departments, Fibers & textiles, chemicals & polymers, construction materials, electronic materials & devices, medical devices [Customers] Processors, manufacturers, trading companies (→ Response / ← Inquiries, suggestions, requests) / [Final products, homes] [Asahi Kasei Group] Customer support center of each product (Customer Support Center, Hebelian Center, Medicine Consultation Line, etc.) Saran Wrap™ cling film, Ziploc™ bags & containers, Hebel Haus™ homes, pharmaceuticals [Customers] Consumers (→ Response / ← Inquiries, suggestions, requests)](/sustainability/social/quality/images/index-img-07.png) Communication with customers
Communication with customers
